Glaze Reactions
The glaze satiron_ZG_Z3N_Z4A_0 with inlay oribe-woof-PAlkAl-PSiMg
is seen here:
This firing is an "invert" firing, dropping first to 1650 degF, then climbing
back to 1850 degF;
The interaction region, while superficially similar, exhibits features not
previously seen.
As before, the interaction region frames a turquoise spiral, with bands of
brooding glossy black.
The matrix (i.e., background) glaze and inlay glaze overlap minimally before
firing. The width of the
The background glaze is satIron_ZG_Z3N_Z4A_0; the inlay design is glazed with
oribe-woof-PAl-PSiMg.
The design is created by adhering a mask and then applying the main
glaze. After drying,
150 deg F an hour to 250 deg F
300 deg F an hour to 2050 deg F
120 deg F an hour to 2330 deg F with a hold of 30 minutes at 2330 deg F
300 deg F an hour to 1650 deg F then a 3 hr hold at 1650 deg F
400 deg F an hour to 1850 deg F then a 2 hr hold at 1850 deg F
300 deg F an hour to 1750 deg F then a 1 hr hold at 1750 deg F
300 deg F an hour to 1700 deg F then a 3 hr hold at 1700 deg F
K2O 0.14
Al2O3 0.69
SiO2 3.65
molecular percent Silica 64.6%
K2O 0.10
Al2O3 0.35
SiO2 3.29
molecular percent Silica 70.58%
Added:
5.0% Copper Oxide
The glaze interactions in the boundary between matrix glaze and inlay glaze
are dominated by kinetic
In both firings, silica migrates from high concentration in the inlay glaze,
to low concentration
The wrinkles in the surface metallic film of the matrix glaze are the result
of early solidification
The drop to a low temperature changes the relative rate at which various
reactions can take place
The greater width of the interaction region, together with the increase in
distinct micro-crystalline
with a different firing profile.
additionally, this firing is to cone 11. The leopard spots,
while present, are not as prominent, as they were
in the prior firing.
There are white and grey micro-crystalline clusters in the
glassy flow surrounding the interaction region.
Additionally, there are
wrinkles in the surface of the metallic layer atop the surface. The
iron-yellow
streaks so prominent in the prior firing are present only in a
vestigial form.
It stands atop a diffuse gunmetal band, which
here is fragmented as if an ice flow breaking up.
original inlay varied from an eighth of an inch to
three eighths of an inch.
the mask is removed and the open area filled in with a
second glaze, using a bulb syringe
with a needle applicator.
Close up Images of the interaction region
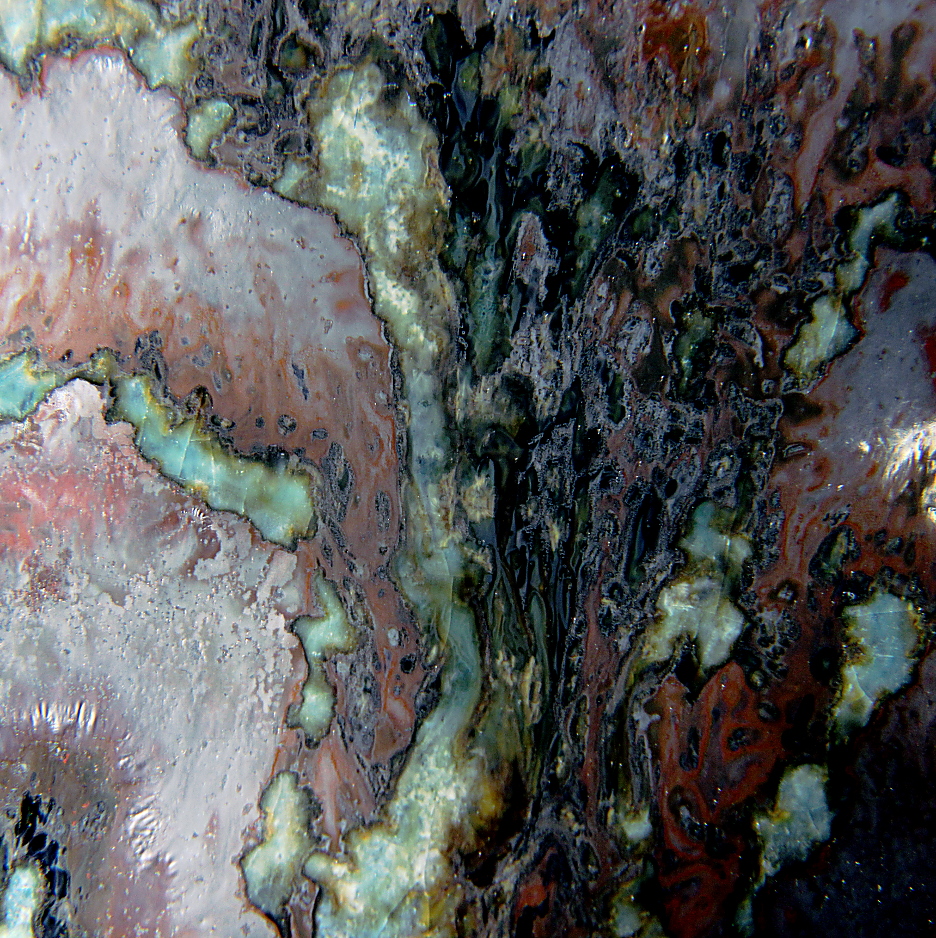



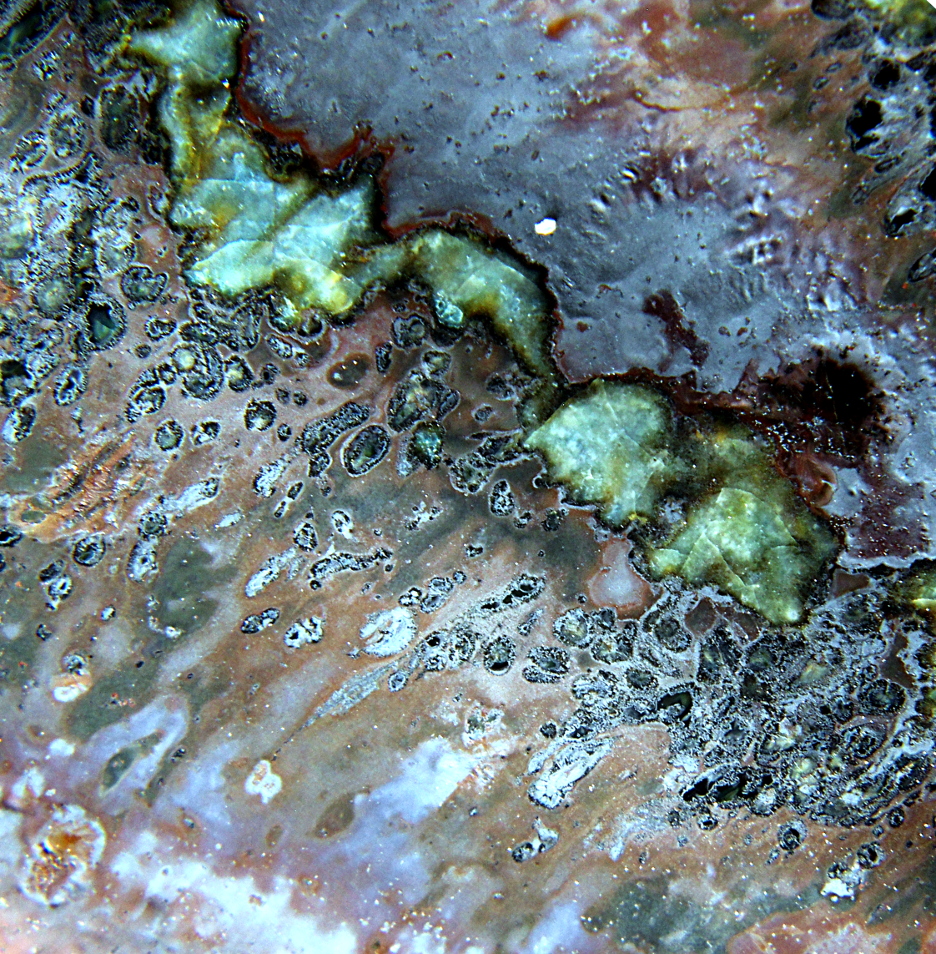
Image of the piece
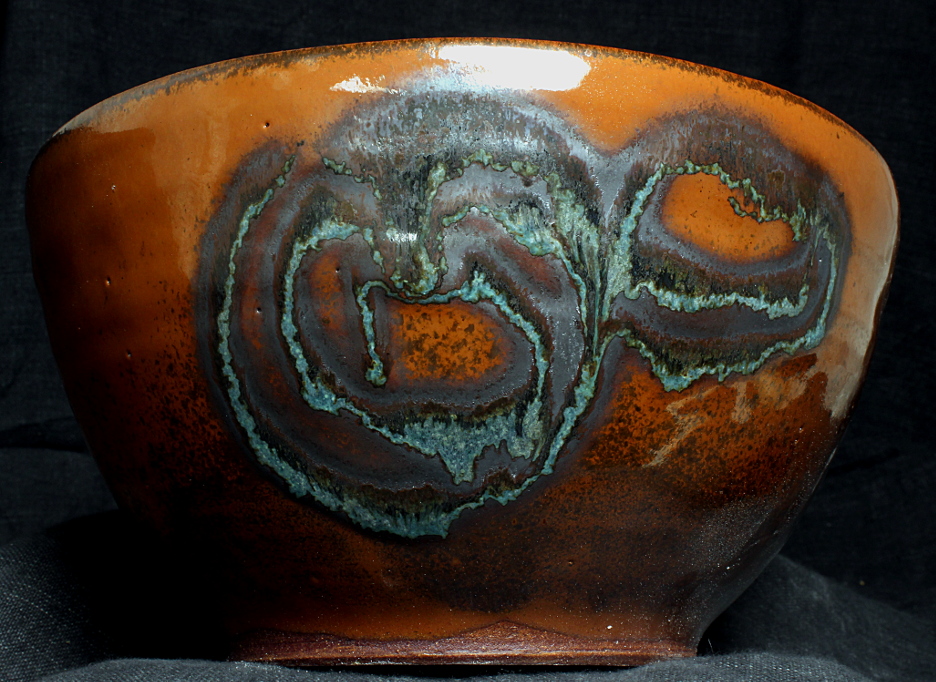
bowl with glaze satIron_ZG_Z3N_Z4A_0 and inlay glaze
oribe-woof-PAlkAl-PSiMg
bowl is ~8.5 inches in diameter
oxidation firing to cone 11 in an electric kiln
Firing profiles
Up Fire profile cone 11
Down Fire Profile cone 11
Clay body is a white stoneware from Clay Art Center in Tacoma, WA.
glaze composition
Empirical Formula satIron_ZG_Z3N_Z4A_0 :
Na2O 0.56
CaO 0.28
MgO 0.02
Fe2O3 0.23
P2O5 0.07
Inlay Glazes:
Empirical Formula oribe-woof-PAl-PSiMg:
Na2O 0.04
CaO 0.61
MgO 0.25
P2O5 0.01
Remarks
effects, i.e these reactions can take place only
as a result of movement of oxides between the matrix
glaze and the inlay
glaze. The various oxides move at different rates, depending on temperature.
in the matrix glaze, likely altering the solubility of
iron in the matrix glaze. This higher solubility of
iron in the matrix glaze
produces the glossy black ring surrounding the inlay.
of the surface film prior to that of the bulk glaze.
As the bulk glaze shrinks, the surface, already solid
can only shrink in
response. The metallic surface film has a rather different composition than
the bulk
glaze. Perhaps the complexity of the firing, down then back up,
allowed this film to grow to a greater
thickness which enhanced the
shrinkage miss-match between surface film and bulk glaze.
and this results in different reactions. Here,
white micro-crystals grew, which did not with the prior firing.
precipitates is the result of more time and
increased variability spent in a region in which the interface
can develop.